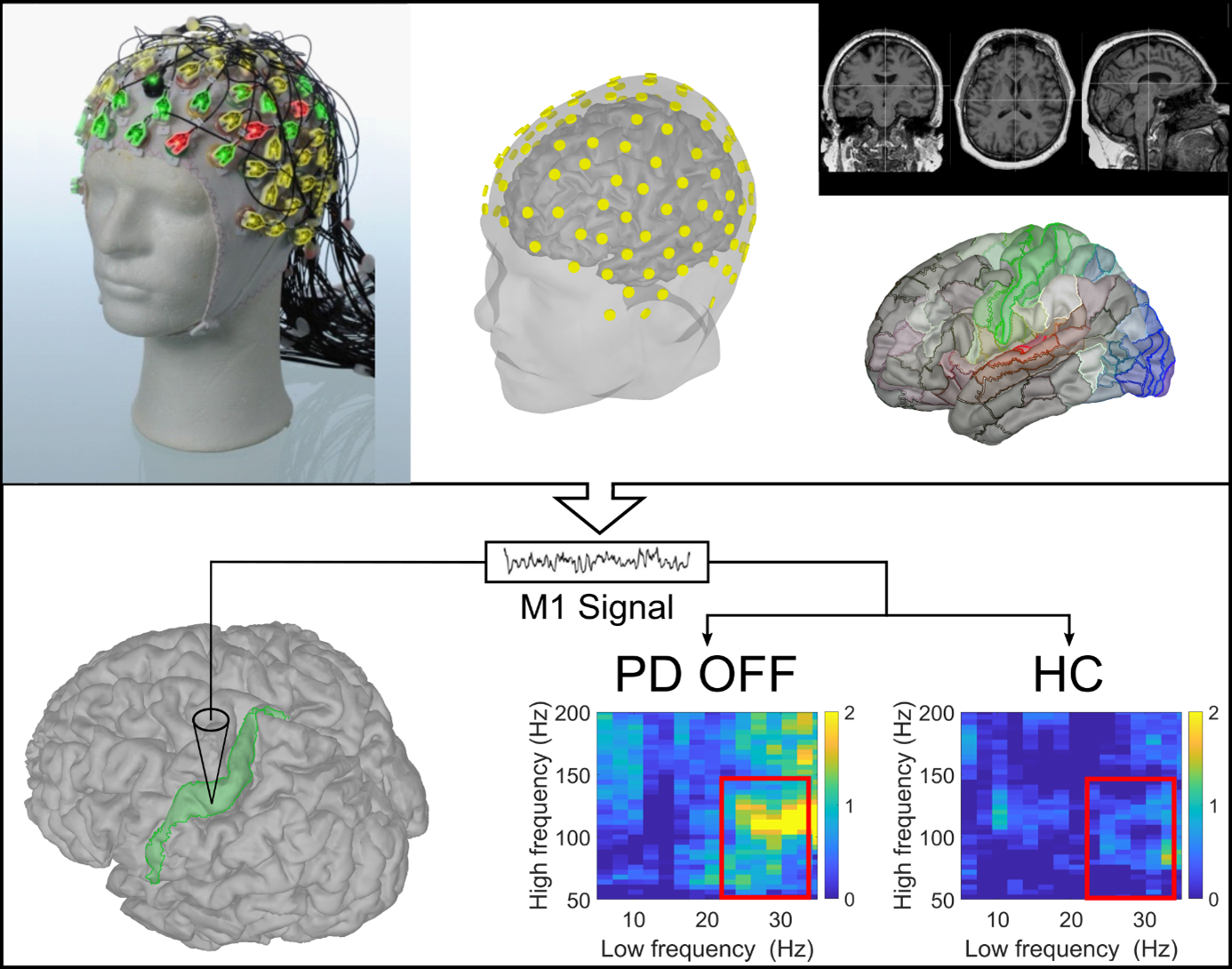
Frequency-specific and spatiotemporal dynamics of β-γ phase-amplitude coupling in Parkinson's disease.
Parkinson's disease disrupts brain rhythms, causing abnormal communication within the motor network that impairs movement. We found a specific pattern of brain rhythms that was abnormally elevated in Parkinson’s and correlated with slowness and stiffness – but not tremor. Medication reduced this pattern, and the reduction was linked to clinical improvement, suggesting it as a non-invasive biomarker and promising target for personalised brain stimulation.
Scientific Abstract
Similar content
Dithering suppresses half-harmonic neural synchronisation to photic stimulation in humans.
Characterising motor and cognitive contributions of cortical beta oscillations and their modulation with rTMS.
Flexible and Stable Cycle-by-Cycle Phase-Locked Deep Brain Stimulation System Targeting Brain Oscillations in the Management of Movement Disorders
Driving theta-gamma oscillations modulates extrasynaptic GABAergic tone: a tACS-TMS study
Frequency-specific and spatiotemporal dynamics of β-γ phase-amplitude coupling in Parkinson's disease.
Parkinson's disease disrupts brain rhythms, causing abnormal communication within the motor network that impairs movement. We found a specific pattern of brain rhythms that was abnormally elevated in Parkinson’s and correlated with slowness and stiffness – but not tremor. Medication reduced this pattern, and the reduction was linked to clinical improvement, suggesting it as a non-invasive biomarker and promising target for personalised brain stimulation.
Scientific Abstract
Citation
DOI
Downloads